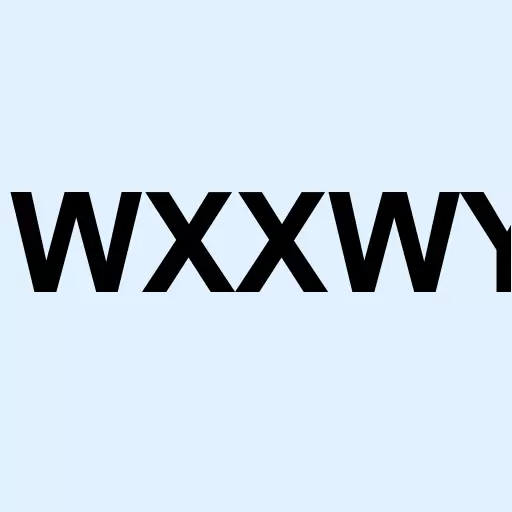WuXi Biologics and Sinorda Biomedicine Enter Strategic Collaboration to Accelerate Development and Manufacturing of Innovative Bispecific Antibody
MWN-AI** Summary
WuXi Biologics and Sinorda Biomedicine have announced a strategic collaboration focused on the development and manufacturing of SND006, a novel bispecific antibody aimed at treating inflammatory bowel disease (IBD) and other autoimmune diseases. This partnership allows Sinorda Biomedicine to leverage WuXi Biologics' extensive experience in biologics to facilitate preclinical studies and accelerate the Investigational New Drug (IND) application process. SND006, independently developed by Sinorda, has successfully completed initial in vitro functional studies, and the company intends to submit IND applications to both China’s National Medical Products Administration (NMPA) and the U.S. Food and Drug Administration (FDA) in 2026.
WuXi Biologics, a leading global Contract Research, Development, and Manufacturing Organization (CRDMO), brings to this collaboration over a decade of expertise in bispecific antibody projects. CEO Dr. Chris Chen expressed optimism about integrating their advanced technology and capabilities to expedite SND006's development, highlighting the importance of making innovative biologics more accessible to patients.
Sinorda Biomedicine, established in 2010, is focused on developing innovative drugs for gastrointestinal ailments and autoimmune diseases. The company is recognized for its unique drug technology platform and is actively expanding its pipeline, which includes various bispecific antibodies under development. Dr. Pingsheng Hu, Chairman of Sinorda, stressed the collaboration's potential to address unmet medical needs and deliver effective therapies globally.
Overall, this strategic alliance signifies a commitment to advancing complex biologics, ultimately aiming to improve treatment options for patients suffering from autoimmune diseases through innovative research and manufacturing processes.
MWN-AI** Analysis
The recent strategic collaboration between WuXi Biologics (2269.HK) and Sinorda Biomedicine for the development and manufacturing of the bispecific antibody SND006 marks a significant advancement in the biopharmaceutical landscape, particularly for treatments targeting autoimmune diseases like inflammatory bowel disease (IBD). As a financial analyst, it is important to consider the implications of this partnership for potential investors and stakeholders in WuXi Biologics.
WuXi Biologics is recognized for its comprehensive CRDMO services and has established a strong reputation in the development of innovative therapeutics. Their extensive experience and technological infrastructure are pivotal to accelerating SND006’s path to clinical trials and FDA approval. Investors should view this collaboration as a strategic opportunity for WuXi Biologics to enhance its portfolio and revenue streams by aligning itself with promising drug candidates.
Sinorda Biomedicine’s commitment to addressing unmet medical needs in autoimmune diseases adds further credibility to this partnership. The completion of in vitro validation studies suggests that SND006 has potential and could become a leading treatment in its category, further benefiting both companies. As regulatory approvals advance, the expectation of increased demand for innovative therapies can boost WuXi’s operational capabilities and market position.
Moreover, WuXi's ongoing commitment to sustainability and ESG practices could enhance its appeal to socially conscious investors, establishing it as a forward-thinking entity in the biopharma sector. Current engagement in nearly 1,000 integrated projects highlights the company's robust operational foundations.
In summary, investors should monitor the developments stemming from this collaboration closely. With the right execution, there is a strong potential for WuXi Biologics to solidify its market standing while paving the way for significant advancements in innovative therapeutics. Thus, maintaining a bullish outlook on WuXi Biologics could be prudent as the partnership progresses.
**MWN-AI Summary and Analysis is based on asking OpenAI to summarize and analyze this news release.
PR Newswire
SHANGHAI, Jan. 28, 2026 /PRNewswire/ -- WuXi Biologics (2269.HK), a leading global Contract Research, Development, and Manufacturing Organization (CRDMO), and Sinorda Biomedicine today jointly announced a strategic collaboration for the development and manufacturing of SND006, a novel bispecific antibody, for the potential treatment of inflammatory bowel disease (IBD) and other autoimmune diseases.
Under the agreement, Sinorda Biomedicine will leverage WuXi Biologics' extensive experience and manufacturing capabilities in biologics development and manufacturing to advance SND006's preclinical pharmacology studies and clinical supply, accelerating the Investigational New Drug (IND) application process. SND006 is an innovative bispecific antibody independently developed by Sinorda Biomedicine, for which the company holds worldwide rights. Sinorda Biomedicine has completed in vitro functional validation studies of SND006 and plans to submit IND applications to both the National Medical Products Administration (NMPA) in China and the U.S. Food and Drug Administration (FDA) in 2026. In the future, the two companies will further expand their collaboration around Sinorda Biomedicine's potential pipeline, including multiple integrated projects spanning from molecule discovery to clinical manufacturing.
Dr. Chris Chen, CEO of WuXi Biologics, commented, "Over the past decade, we have accumulated experience across hundreds of projects in bispecific and multispecific antibodies, which have become one of our fastest?growing areas. We are pleased to accelerate the development and manufacturing of Sinorda Biomedicine's innovative bispecific antibody SND006 through our integrated technology platforms and comprehensive capabilities. Looking ahead, we will continue accelerating and transforming biologics discovery, development and manufacturing to empower global partners and make innovative biologics more accessible and affordable for patients worldwide."
Dr. Pingsheng Hu, Chairman and General Manager of Sinorda Biomedicine, commented, "SND006 is a potentially best-in-class innovative bispecific antibody discovered and developed by Sinorda Biomedicine, with the potential to deliver breakthroughs in the treatment of gastrointestinal and multiple autoimmune diseases. WuXi Biologics is a global leader in CRDMO services, offering truly end?to?end solutions underpinned by accumulated know-how, comprehensive technology platforms, and a strong track record—particularly in the development and manufacturing of bispecific antibodies. We believe this collaboration will accelerate the IND filings of our innovative biologics in China and worldwide, address unmet medical needs in autoimmune diseases, and ultimately bring safe and effective therapies to patients."
About WuXi Biologics
WuXi Biologics (stock code: 2269.HK) is a leading global Contract Research, Development and Manufacturing Organization (CRDMO) offering end-to-end solutions that enable partners to discover, develop and manufacture biologics – from concept to commercialization – for the benefit of patients worldwide.
With over 12,000 skilled employees in China, the United States, Ireland, Germany and Singapore, WuXi Biologics leverages its technologies and expertise to provide customers with efficient and cost-effective biologics discovery, development and manufacturing solutions. As of December 31, 2025, WuXi Biologics is supporting 945 integrated client projects, including 74 in Phase III and 25 in commercial manufacturing.
WuXi Biologics regards sustainability as the cornerstone of long-term business growth. The company continuously drives green technology innovations to offer advanced end-to-end Green CRDMO solutions for its global partners while consistently achieving excellence in Environment, Social and Governance (ESG). Committed to creating shared value, it collaborates with all stakeholders to foster positive social and environmental impacts and promote responsible practices that empower the entire value chain.
For more information about WuXi Biologics, please visit: www.wuxibiologics.com.
About Sinorda Biomedicine
Sinorda Biomedicine was established in 2010 and is a biomedical innovation company in the commercialization stage. The company focuses on the research and development and industrialization of innovative drugs for digestive tract diseases, tumor immunity and autoimmune diseases. It has a bioinnovative drug technology platform, a clinical medical R&D platform and an experienced international R&D team, with successful experience in innovative drug applications in China, Europe, America and other countries.
Linaprazan glureta (X842), the company's self-developed class 1.1 new drug for treatment of gastric acid-related diseases, has been successfully approved for commercialization and industrialization. The sentinel lymph node T cell project for solid tumor treatment has clinical IIT research results for various tumors. In addition, the company has a number of early-stage product pipelines, including bispecific antibodies for the treatment of autoimmune disease IBD.
The company has developed rapidly through extensive cooperation with domestic and foreign pharmaceutical companies and R&D institutions, and is committed to becoming the most valuable innovative pharmaceutical enterprise in China.
WuXi Biologics Contacts
Business
info@wuxibiologics.com
Media
PR@wuxibiologics.com
SOURCE WuXi Biologics
FAQ**
How will the collaboration between WuXi Biologics Cayman Inc ADR WXXWY and Sinorda Biomedicine impact the speed and efficiency of the IND application process for the bispecific antibody SND006 aimed at treating inflammatory bowel disease and autoimmune diseases?
Considering WuXi Biologics Cayman Inc ADR WXXWY's extensive experience in bispecific antibodies, what specific technologies or platforms will be employed to expedite the development and manufacturing of SND006 in this strategic collaboration?
What are the potential implications for WuXi Biologics Cayman Inc ADR WXXWY’s financial performance as they partner with Sinorda Biomedicine to advance SND006 and possibly expand their collaboration to include additional projects in Sinorda's pipeline?
In what ways will the partnership between WuXi Biologics Cayman Inc ADR WXXWY and Sinorda Biomedicine contribute to addressing the unmet medical needs in autoimmune diseases, and how do both companies plan to ensure patient access to the innovative therapies developed through this collaboration?
**MWN-AI FAQ is based on asking OpenAI questions about Wuxi Biologics Cayman Inc ADR (OTC: WXXWY).
NASDAQ: WXXWY
WXXWY Trading
-3.38% G/L:
$8.792 Last:
11,387 Volume:
$8.96 Open: